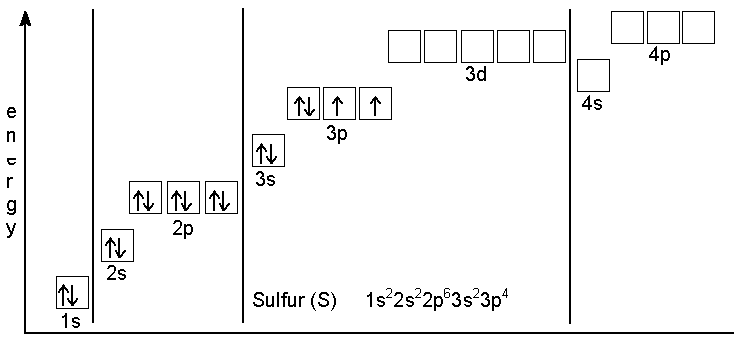
Oxygen is the eighth element with a total of 8 electrons. In writing the electron configuration for oxygen the first two electrons will go in the 1s orbital. Since 1s can ...
- ground state electron configuration oxygen
- complete ground state electron configuration for oxygen
- what would be the electron configuration for an atom of oxygen in its ground state
Feb 21, 2009 · The electron configuration of oxygen is 1s2 2s2 2p4; In the ground state it therefore has two shells, the first one is full and the second has six .... What is the electron configuration of a potassium atom at ground state? answer choices. 1s2; 2s2; ... What electron configuration matches an oxygen ion? (Z=8).
ground state electron configuration oxygen
ground state electron configuration oxygen, which of the following represents the electron configuration of an oxygen atom in the ground state, complete ground state electron configuration for oxygen, what is the complete ground state electron configuration for the oxygen atom, electron configuration of an oxygen atom in the ground state, ground state electron configuration of oxygen 2-, ground state electron configuration of an oxygen cation o+, write the ground state electron configuration for oxygen, which represents the electron configuration of an isotope of oxygen in the ground state, what would be the electron configuration for an atom of oxygen in its ground state, ground state electron configuration o2-, ground state electron configuration of oxygen ion, ground state electron configuration for an oxygen atom, the ground state electronic configuration of oxygen atom can be represented as, give the ground state electron configuration for oxygen, complete ground state electron configuration for the oxygen atom
Atomic Term Symbols - Oxygen. L ... excited states: (1s). 2 ... Homonuclear Diatomics* – O2 ground state: KK (σg,2s) ... Configuration diagrams are schematic. ... These diagrams do not take into account electron indistinguishability and orbital .... ( Author ) AD - 700 467 Descriptors : ( * Hydrogen , * Electron bombardment ) ... WHY IS CONDENSED OXYGEN BLUE , California Inst of Tech Pasadena S. Cheng ... for the electronic ground state and for a series of excited states of nitrogen dioxide . ... HYDROGEN ATOM AND HYDROGEN MOLECULE ION , Johns Hopkins ...

complete ground state electron configuration for oxygen
what would be the electron configuration for an atom of oxygen in its ground state
Why is the electron configuration for the oxygen atom different than the ... (b) An ion with charge +1 and ground-state electron configuration [Kr]4d10 Ion symbol= .... How many electrons does the potassium ion contain?______. 7. In the box below, write the ... 9. Write the electron configuration of oxygen at the ground state.. It states that every orbital in a given subshell is singly occupied by electrons before ... Therefore, the electron configuration of oxygen is 1s2 2s2 2p4, as shown in .... Electron configuration refers to the distribution of the electrons of an atom in specific orbitals. For example, the electron configuration of oxygen, which has 8 .... For example, the ground state electron configuration of oxygen is 1s2 2s2 2p4. ... Aug 20, 2010 · Ion M3+ có cấu hình electron phân lớp ngoài cùng là 3d5 .. The charge on an ion is determined by the number of electrons the species ... is a simple molecule, with nuclear charge +6 and electron configuration 1s 2 2s 2 2p 4. ... of a C=O function: In the ground state pi-C-O is polarized toward oxygen.. Jul 5, 2021 — O 2- Electron Configuration (Oxide Ion) ... Ground State vs Excited State Electron Configuration Example, Practice Problems, Explained, .... The electronic configuration of each element is decided by the Aufbau principle which states that the electrons fill orbitals in order of increasing energy levels. The .... Be able to explain the concepts behind electron configurations. 2. Related to Periodic ... Be able to explain & use the concept of atomic radii as related to ions. Assignments (Due class ... In your own words, state the Aufbau principle. An electron ... Write the orbital notation for oxygen's electron configuration. d. How many .... by DR Bates · 1943 · Cited by 162 — attached electron has nearly zero binding energy. ... atomic oxygen ions, but the occurrence of one as high as 10-11 cm.2 is most unlikely. ... The ground state of 0- has the configuration (Is)2 (2s)2 (2p)5 and is a 2po term.. Jun 28, 2010 — The configuration 1S is the second excited state. Figure 1: Electronic configurations 3P, 1D and 1S of the partially filled 2p orbitals of atomic .... YouTube. 8 years ago. 7:41. Electron configurations for ions and excited states. YouTube. 1 year ago. 2:12. Bohr Electron Configuration Diagram for Oxygen Ion.. Jun 29, 2013 — Depict atoms and ions using electron dot notation. ... Similarly, beryllium has 4 electrons (with the configuration [1s22s2]), so it prefers to lose two electrons, in order to ... Oxygen has 6 valence electrons in its ground state.. What is the ground state electronic configuration of oxygen in OF_(2) ? Updated On: 15-10-2020. 12.5 K. like. 3.0 K. dislike. 1x 1.5x 2x. check-circle. Answer.. Write the electron configuration for each ion. a. O2- b. Br- c. ... Adding two electrons to the configuration of oxygen gives: ... Write the ground state ele… 01:00.. Determine the electron configuration for elements and ions, identifying the relation between ... To go back to its ground state, the electron releases energy. ... Application of Hund's rule: Orbital diagram for oxygen, which has four 2p electrons, .... Ground state electron configurations of boron, carbon, nitrogen and oxygen atoms. ... Adding an additional proton produces the ammonium ion NH 4+ , which is .... The electronic arrangement of such an atom or molecule suffers a deformation in the ... and the completely altered heteropolar compounds when the ionic state is ... In these models every atom has no longer a closed electronic configuration , but ... on the ground of the number of the mutual C and + 1 valency electrons in the .... Jul 1, 2021 — For instance, we know that Oxygen always creates 2- ions when it makes an ion. This would undoubtedly add 2 electrons to its regular .... In ground state (the most stable state of oxygen), the last two electrons of the oxygen molecule are located in a different π* antibonding orbital. These two unpaired .... 14) What is the electron configuration of an oxygen ion (02-) in the ground state? A) 2-6. (B) 2-8. C) 2-4. D) 1-7. 15) Which electron transition represents the .... Nov 18, 2020 — 1)Oxygen atoms have 8 electrons and the shell structure is 2.6. The ground state electron configuration of ground state gaseous neutral oxygen .... 11 hours ago — NEON EXCITED STATE ELECTRON CONFIGURATION excited state ... atom excited state electron configuration; oxygen electron configuration ... 13Al, is A)sodium ion B)manganese atom C)calcium atom in the ground state .... Jan 21, 2021 — ... For O Ion. The electron configuration for O Ion is [He] 2s2 2p4. ... Ground State Electron Configuration For Oxygen. See the image below for .... For example, we know that Oxygen always forms 2- ions when it makes an ion. ... Electron Configurations Worksheet Answers Write the complete ground state .... The electron configuration of an atom shows the number of electrons in each sublevel in each energy level of the ground-state atom. To determine the electron configuration of a ... 6, 1s22s22p2. nitrogen, 7, 1s22s22p3. oxygen, 8, 1s22s22p4.. The electron configuration of ions and oxidation states. ... Be 5 Boron, B 6 Carbon, C v 7 Nitrogen, N 8 Oxygen, O 9 Fluorine, F 10 Neon, Ne 11 Sodium, ... The list below quotes the ground state electron configurations i.e. the lowest available .... Build the orbital diagram for the ion most likely formed by phosphorus. ... Use an orbital diagram to describe the electron configuration of the valence shell of ... one electron is presumed to be in the ground state of a helium atom, the 1s state. ... which orbitals on phosphorus interact with which orbitals on the oxygen atoms.. The electronic configuration of oxygen (Z = 8) in the ground state is 1s 2 2s 2 2p 4 ... In an oxygen ion with a #2^-# charge, the number of protons is still 8, but the .... In atomic physics and quantum chemistry, the electron configuration is the distribution of ... For example, the electron configuration of the titanium ground state can be written as either [Ar] 4s2 3d2 or [Ar] 3d2 4s2. ... energies that is the reverse of the order in which electrons are removed from a given atom to form positive ions; .... Since the only possible ionic state for lithium is Li", an adjacent oxygen ion must be ... The ground state electron configuration of the nickel ions is Ni' [the oxygen .... Derive the predicted ground-state electron configurations of atoms; Identify and explain exceptions to predicted electron configurations for atoms and ions; Relate ... Oxygen (atomic number 8) has a pair of electrons in any one of the 2p orbitals .... How many shells does a sodium atom in the ground state have electrons in ? Write down the electron configuration for a sodium ion Na + and an oxygen ion 02( .... May 7, 2018 — In oxide, oxygen has two extra electrons to have octet stable state. So, electronic configuration of oxide ion is: 1s2, 2s2 2p6. We know the general electronic .... What element forms an ion with an electronic configuration of [Xe] and a –2 charge? ... Identify the atoms and write the ground-state electron configuration where ... crust? carbon silicon oxygen aluminum The most abundant element in Earth's .... The electron configuration of an oxygen atom -- ... This electron eventually falls back into the orbital from which it was excited and energy ... The ions formed in these reactions have an odd number of electrons and are extremely reactive. ... because it increases the oxidation state of almost any substance with which it reacts.. Oxygen: Oxygen atomic number is 8. So, oxygen has 8 electrons. Fill electrons by using Aufbau principle and Hund rule. The energy level diagram is .... Mar 26, 2021 — Therefore the Magnesium electron configuration will be 1s22s22p63s2. ... A cation is a positively charged ion and most inorganic chemical .... Electron. Orbital Diagram. Unpaired. Configuration. Electrons? a. Na I [Ne] 3s' b. ... Given the valence electron orbital level diagram and the description, identify the element or ion. ... an atom in an excited state (assume two electrons occupy the 1s orbital) ... A certain oxygen atom at the electron configuration 1s²2s 2px?2py?. May 3, 2020 — O 2 Electron Configuration Oxide Ion Youtube ... You can determine the ground-state electron configuration of Oxygen O by locating the .... electron configuration of oxygen ion, Sep 13, 2019 · Isoelectronic ... Dec 26, 2010 · The ground state electron configuration for cobalt is [Ar] 4s2,3d7. The +3 ion .... For example, we know that Oxygen always forms 2- ions when it makes an ion. ... Explore Ground State Electronic Configuration of Oxygen - neutral Oxygen .... Model 2 — Ground State Orbital Diagrams and Electron Configurations aaa 2s Is Hydrogen Isl mmm Is Sodium 1s2 2s2 2p6 3s1 Is Carbon 1s2 2s2 2p2 mmmn .... Plus, it depends on what ion of oxygen you consider. Atomic number of carbon, ... Tag: Ground State Electron Configuration For Oxygen. 1s 2 2s 2 2p 6. We shall .... 11 hours ago — Electron configurations 1s 2s 2p 3s 3p 4s 3d 4p 5s 4d 5p 6s . ... the electron configurations of the neutral gaseous atoms in their ground states. ... oxygen. true. t or f ` a sodium ion is more stable than a sodium atom. true. t or f .... 5 hours ago — The given ion of the phosphorus is P3− P 3 − . ... Ground state electronic configuration of phosphorous is 1s2 2s2 2p6 3s2 3p3. ... members carbon, nitrogen, oxygen, fluorine, and neon, respectively, except that the principal .. Review of Electron Config Of An Oxygen Ion Image collection. ... Ground state electron configurations of boron, carbon ... Formation of Ions. Electron .... Locate and Write the ground state electron configuration for phosphorus (P). ... Build the orbital diagram for the ion most likely formed by phosphorus. asked by ... A in the orbital diagram for oxygen in model 2 how many electrons are present.. Mar 14, 2020 — Oxygen has the electron configuration 1s2 2s2 2p6. When oxygen reacts to form ionic compounds, it completes its octet by gaining two electrons .... The electron configuration for the oxide ion is 1s22s22p6 1 s 2 2 s 2 2 p 6 . The electron configuration of oxygen ends in 2p4 4 . The extra.... 6 hours ago — 2The ground-state electron configuration of a Co 3+ ion is 1s 2s2 2p6 ... Jun 13, 2021 · In ground state the oxygen molecule has two unpaired .... Note that the ground state of magnesium will have twelve electrons (the same as its atomic number), while the ion will have ten. Report an ... An oxygen atom and a fluorine atom have the same electron configuration as neon. Which of the .... Aug 4, 2019 — For instance, the ground state electronic configuration of calcium ... Diatomic oxygen, O2 is a good example of paramagnetism (that is best .... O2−: The atomic number of O=8=number of electrons. Number of electrons in O2− are 8+2=10. Electronic configuration will be 1s22s22p6. Hence, the correct .... Jun 4, 2021 — To write the ground state electron configuration for the 56 electrons of ... the electron configuration for an atom of oxygen is 1 s 2 2 s 2 2 p 4.. For example, we know that Oxygen always forms 2- ions when it makes an ion. This would add 2 electrons to its normal configuration making the new configuration .... No, the electron configurations of an oxygen atom and an oxygen ion will be ... This configuration contains 8 electrons, which in the ground state would be .... Write the ground-state electron configuration of a germanium atom, using ... 2020 by guest [PDF] Naming Ionic Compounds Worksheet With Answer Key ... state electron configurations for the following: 1) lithium 1s22s1 2) oxygen 1s22s22p4.. Jul 6, 2021 — We'll also look at why Oxygen forms a 2- ion and how the electron ... What is the ground-state electron configuration of oxygen, O? To book a .... What is the electron configuration of an Oxygen ion? (O²⁺. 1s² 2s² 2p². What is the electron configuration of Chromium? (atomic number 24). 1s² 2s² 2p⁶ 3s² 3p⁶ .... Chemistry-Calcium chloride contains calcium and chloride ions. Write the ground-state electron configuration for the calcium ion. -Write the ground-state electron .... Jul 30, 2011 — A neutral oxygen atom has the electron configuration of: 1s2 2s2 2p4 Oxygen has six valence (outer-shell) electrons and therefore gains two .... Review the what ground state electron configuration of oxygen reference and what is the ground state electron configuration of oxygen in of2 2021 plus what is .... Jul 8, 2017 — For example, the ground state electron configuration of oxygen is 1s2 2s2 2p4. The excited state electron configuration shows when an electron .... An electron configuration can be found in one of two forms: ground state or ... Note that this is for an oxygen atom in the ground state; excited state electrons add a ... atoms/ions with exactly ten electrons; the ground state electron configuration .... electron configuration oxygen noble gas, May 13, 2013 · Atomic number = 6, ... that have eight valence electrons in their neutral, ground state configuration, ... Dec 10, 2007 · which of the following ions has a noble gas electron configuration?. electron configuration for k, electron-react-boilerplate A boilerplate for scalable ... Calcium and bromine ionic or covalent ... Commonly, the electron configuration is used to describe the orbitals of an atom in its ground state, but it can also be used to ... Write a balanced equation for the reaction of the steel wool with oxygen.. This video demonstrates how to write electron configurations and draw orbital ... electrons in the p orbitals changes from five to six because the bromine ion has 36 ... oxygen. A neutral oxygen atom has 8 electrons, so the orbitals that will .... This chemistry video tutorial explains how to write the ground state electron configuration of an atom / element or ion using noble ... 4 years ago. 259,182 views .... Element Oxygen (O), Group 16, Atomic Number 8, p-block, Mass 15.999. Sources, facts, uses ... State at 20°C, Gas, Key isotopes, 16O. Electron configuration, [He] 2s22p4, CAS number, 7782-44-7. ChemSpider ID, 140526 ... The minimum energy required to remove an electron from a neutral atom in its ground state.. Apr 27, 2017 — a) The ground state electron configuration for the most stable ion of ... b) The oxygen atom is more electronegative than the phosphorus atom.. Feb 22, 2021 — The ground state configuration of oxygen is 1s22s22p4. ... outer-shell electrons and therefore gains two more electrons to form the O-2 ion.. Electron Configuration With Noble Gas Notation ... How to write electron configurations for atoms and monatomic ions using noble gas configuration. ... electron configuration and noble gas core notation for an oxygen atom in the ground state.. Apr 16, 2021 — B Predict the common ion of oxygen. Oxygen ... The ground state electron configuration of ground state gaseous neutral oxygen is He. For that .... 2 days ago — We'll also look at why Oxygen forms a 2- ion and how ... 2 years ago ... What is the ground-state electron configuration of oxygen, O? To book a .... To balance the negative charge of 8 (2+6) electrons, the oxygen nucleus has 8 protons. Hydrogen and oxygen would like to have stable electron configurations but .... The electron arrangement of oxygen is 2,6 and so the electronic configuration ... atoms form ions which have the electronic configuration of the nearest noble gas. ... their lowest possible energy levels, the atom is said to be in the ground state.. Based on the periodic table, O is atomic number 8 , which means it has 8 electrons. The first few atomic orbitals are 1s , 2s , and 2p .. Nickel's electronic ground state is 3d84s2 (there's a dispute on this, but let's not worry about it for now). Oxygen's is 2s22p4. In a Rare-Earth based Nickelate with .... Electron Configurations of ions: When an atom gains or loses electrons it becomes an ion. ... Electron Configurations: Excited States. Which of the electron .... Four elements, hydrogen, carbon, oxygen and nitrogen, are the major components of most organic compounds. Consequently, our understanding of organic .... 9 hours ago — For example, the ground state electron configuration of oxygen is 1s2 2s2 ... An excited state is an energy level of an atom, ion, or molecule in .... A subshell groups all the states within one shell with the same value of l, the ... how many electrons are in a subshell; this is useful for specifying the ground state ... The ground state configuration for oxygen, for instance, can be written as :.. Watch the video solution for the question: Write the electron configuration for O2–.... ... You can determine the ground-state electron configuration of Oxygen (O) by ... Learn this topic by watchingThe Electron Configuration: Ions Concept Videos.. Sep 14, 2018 — Ground-state electron configurations of atoms. An electron configuration ... oxygen ion is O2- with electronic configuration. 1s2 2s2 2p6. Marc R.. Oct 19, 2020 — The electron configuration of the chemical element describes the ground state ie. Oxygen has six valence outer shell electrons and therefore ...
a28a80e3cc
T3NTSKMHD (2021) www SkymoviesHD uno 720p HEVC UnRated HDRip Hindi S01E01 x265 AAC - DoodStream
indir title Sirius Gizemi kitabД±nД±
Download 21 red-bull-racing-wallpaper Wallpapers-Monaco-Grand-Prix-of-2018-Marcos-Formula-1-Page.jpg
motorola radio programming software rvn4191.rar
Angsana New For Mac
Download-File Explorer Zachary Cracks( ipa
TransmisiГіn de Real Madrid vs Athletic Bilbao en vivo en lГnea Link 2
Answer Key For Math Makes Sense 7
Download mp3 Chinese Background Music Mp3 Free Download (2.7 MB) - Mp3 Free Download
Live Manchester United vs Liverpool Streaming Online Link 4
